Boil off Calculator
Calculate the effects of evaporation on wort gravity and volume.
Gravity and volume changes due to the effects of evaporation. When water boils, only the water evaporates, leaving a smaller, more concentrated quantity of liquid behind. We can estimate the amount of sugar in this liquid if we have an idea about how quickly our evaporation occurs.
Input
As usual, the lighter coloured boxes are your input boxes, the grey boxes are our calculation results.
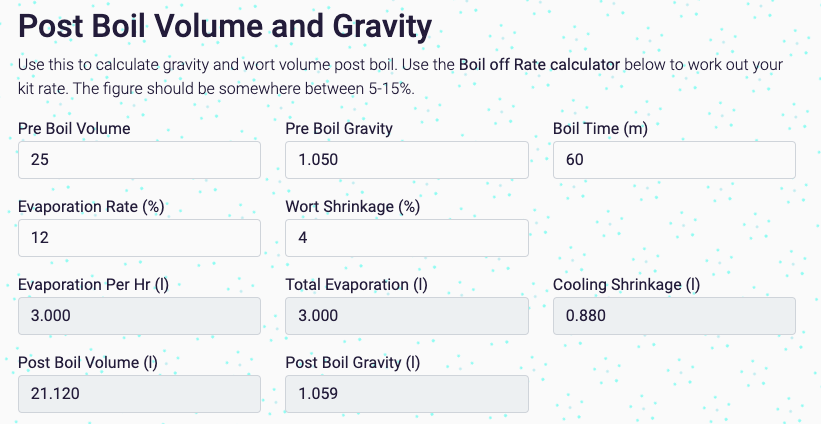
- Pre Boil Volume: the starting volume.
- Pre Boil Gravity: your starting gravity.
- Boil Time: how long you're boiling for.
- Evaporation Rate: rate (percentage per hour) of evaporation. You can test this in the Boil off Rate calculator below.
- Wort Shrinkage: how much your wort shrinks due to cooling.
Results
Once you've added the figures above, you'll get your results.
- Evaporation per Hour: the number of litres from your given volume lost per hour of boiling.
- Total Evaporation: total number of litres lost due to evaporation
- Cooling Shrinkage: number of litres lost due to shrinkage
- Post Boil Volume: volume of wort after boil/cooling.
- Post Boil Gravity: gravity of wort after boil/cooling.

This calculator will give you your Evaporation Rate for use in the Post Boil Volume and Gravity calculator.
You'll need to be able to measure the volume in your kettle accurately. You may have a sight on your kettle, or potentially you could use a measuring stick to calibrate it. You could do this with just some water for an hour, or on brewday. Just record the volume before and after the boil.
If you want to enter a volume that has been measured at 100C or just off boiling, you can change the wort shrinkage to 0.

This is an alternative way to estimate boil off. Larger diameter vessels will boil off more water, but this really depends on how much power you're putting into the water. Higher kW = more evaporation.
Adjust the power factor figure to tweak it till it has the correct amount.
todo
- [ ] Save evaporation rate to user preferences
James Torr 18 Jun 22